Genetic Link Found Between Rare Bone Disease and Meniere's
A gene fault that leads to soft bones and developmental issues also may lead to a form of bilateral Meniere's Disease found more often in men
Over the past six months I have had the great pleasure to interview and write about the work of Antonio Lopez-Escamez who is looking at, amongst other things, potential genetic links to Meniere’s disease, as well as the work of Andreas Eckhard and David Bächinger who found that one subgroup of Meniere’s patients seem to have a poorly developed part of the inner ear. (If you have not read about the work of these scientists, links are below)
So it is of course hugely exciting to be able to bring you news of a recently published paper that describes work from all three of these great Meniere’s researchers (alongside others). This study demonstrates quite conclusively that a fault in a gene that causes a rare issue with bone formation also makes it much more likely that the endolymphatic sac in the inner ear will not develop properly which in turn can lead to Meniere’s disease. It is worth saying from the outset that this gene fault is very rare, so this is not a discovery that has implications for most Meniere’s sufferers, but it is exciting confirmation of one genetic route by which Meniere’s can occur.
The paper is available here. The disease that the investigators examined is called “X-Linked Hypophosphatemia” (XLH). The last part means literally “Low (hypo) phosphate in the blood (-emia)”.
To understand what X-linked means requires us to remember some basic genetics. Every cell in our bodies contains genes, the instructions for making all the proteins needed to build and run a human. Those genes are gathered into packages called chromosomes, and we have 23 pairs of packages in most cells.
One package in each pair comes from mum and one from dad, so although these two copies contain the instructions for making certain proteins, the copy from each parent might be slightly different. This pairing makes genes robust - if there is a mutation in an essential gene in one of the pairs, chances are the other will be ok. One pair though is special, as they determine the sex of the person. Chromosome pair 23 consists of some combination of two chromosomes that are not the same, one shaped like a “X” and one like a “Y”. Females end up having two copies of the X - one from each parent, whereas males will have one X and one Y. The X and Y chromosomes do not contain the same genes - which is one reason males and females develop differently. This also means that males are at a disadvantage - if there are mutations in their X chromosome, there is no backup copy. This is what X-linked means - a condition that stems from changes in the X chromosome, making that change much more likely to have an impact on males.
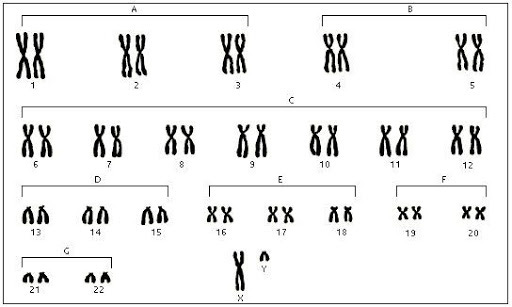
So we have a disease caused by a genetic change to a gene called “PHEX” on the X-chromosome that is therefore more likely to impact males and leads to a shortage of an essential chemical - phosphate - in the blood. If the genetic change totally breaks the gene so that it no longer does any of what it should, it is called a “loss-of-function” mutation. Phosphate, along with calcium, is required to build solid bone - these substances convert the collagen mesh that initially forms as bone grows into rigid strong bone. Lack of phosphate leads to soft bones, risk of stress fractures, dental issues and arthritis in adults, and in children bow-leggedness, short stature and issues walking that are sometimes called “rickets”.
What did the researchers do?
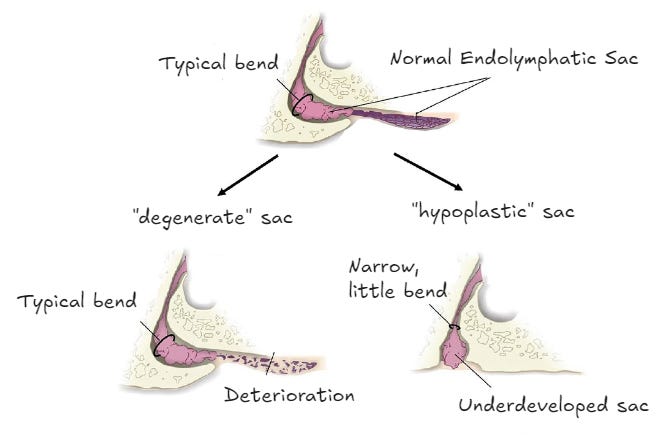
The work of Andreas Eckhard and David Bächinger that I previously covered showed that a subgroup of about 30% of Meniere’s patients have a characteristic deformity of the inner ear. Their endolymphatic sac has not properly developed (termed “hypoplastic”). This can be seen by checking the bone structure of the skull from scans of the head. People with this issue tend to be men and to develop bilateral Meniere’s more often. (The other 70% of Meniere’s sufferers have a sac that has deteriorated or “degenerated” for some reason). The researchers suspected that maybe there was a connection between the XLH genetic disorder and this under development of the endolymphatic sac.
They recruited 33 people with XLH and examined them to see which had Meniere’s and of those, which appeared to have the under developed endolymphatic sac. Meniere’s is thought to impact around 2 out of every 1000 of the population, and only 30% of those will be in the "hypoplastic” subgroup we are interested in. XLH is even rarer, at around 5 per 100,000 people. So the chances of someone having both at random if they were not connected is a tiny 1 in 33 million. And yet of the 10 men and 23 women with XLH in the study, 6 had Meniere’s and showed an underdeveloped sac. This is incredibly unlikely to be down to chance alone, and is a strong indication of some kind of connection between the gene mutation and this form of Meniere’s. All were men, which fits with the previous work on hypoplastic sacs in Meniere’s.
The “Gene Dosage” idea
There is some important nuance that I have so far missed out in order to simplify the explanation. In females, who have two copies of the X chromosome, which one gets used? The answer is that each cell seems to randomly switch off one or other of them, meaning that some cells get the faulty PHEX gene and some get the working one. This “mosaic” pattern means that although females can suffer symptoms from the PHEX gene they are generally not so bad. This study found exactly that, with those women that had Meniere’s suffering less severe vertigo and structural damage. It is also possible for the PHEX gene to be faulty but not entirely broken. In that case, it would still party do its job. This explains why there were also men who had Meniere’s but with less severe symptoms.
What this means for the future
This is the first study to link the PHEX gene to inner ear issues that can drive development of Meniere’s. It strongly suggests that men with fully broken PHEX genes are at a very high risk of developing bilateral Meniere’s disease. This means that early genetic testing combined with scans to look for poorly developed endolymphatic sacs can identify Meniere’s risk much earlier, before hearing loss or balance issues begin occurring. This in turn gives the possibility that targeted treatments may be able to prevent development of Meniere’s in these people. Just one example of the way that advances in genetic testing aligned with good quality science is pushing our understanding of Meniere’s ever further.
huge thanks to David Bächinger, one of the authors of the research, who checked the article for me.
Additional Resources
NIH Intro to Chromosomes - https://www.genome.gov/genetics-glossary/Chromosome
Wikipedia on SLH - https://en.wikipedia.org/wiki/X-linked_hypophosphatemia
Image attribution
NIH (Public domain)
23 chromosomes ZBY360, CC BY-SA 4.0
Modified from https://www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2025.1607435/full CC BY license
AI Statement
No AI was used to create the text in this article. AI was used at times to help with some of the research, but sources were always followed up and no AI output was used in the final text. AI was used to help generate the some of the images and thumbnail




Thank you for another informative piece, always worth a read!